⊗This PRODUCT IS INTENDED AS A RESEARCH CHEMICAL ONLY. This designation allows the use of research chemicals strictly for in vitro testing and laboratory experimentation only. All product information available on this website is for educational purposes only. Bodily introduction of any kind into humans or animals is strictly prohibited by law. This product should only be handled by licensed, qualified professionals. This product is not a drug, food, or cosmetic and may not be misbranded, misused or mislabeled as a drug, food, or cosmetic.
⊗This PRODUCT IS INTENDED AS A RESEARCH CHEMICAL ONLY. This designation allows the use of research chemicals strictly for in vitro testing and laboratory experimentation only. All product information available on this website is for educational purposes only. Bodily introduction of any kind into humans or animals is strictly prohibited by law. This product should only be handled by licensed, qualified professionals. This product is not a drug, food, or cosmetic and may not be misbranded, misused or mislabeled as a drug, food, or cosmetic.
BAM15 50mg
60 Capsules
BAM15 is a selective mitochondrial uncoupler that enhances metabolic efficiency by disrupting the coupling between electron transport and ATP synthesis in mitochondria. Research demonstrates BAM15 significantly increases energy expenditure, promotes fat loss while preserving lean muscle mass, and improves insulin sensitivity. In animal studies, BAM15 prevented diet-induced obesity, decreased fat mass by 20-30%, reversed insulin resistance, and improved metabolic markers without altering body temperature or food intake. The compound targets lipid-rich tissues, reduces hepatic steatosis, and activates AMPK-mediated fatty acid oxidation. Studies show BAM15 preserves muscle during weight loss, enhances mitochondrial function, and reduces inflammation through NLRP3 inflammasome inhibition.
Peptides
What is Bam15?
BAM15 (N5,N6-bis(2-Fluorophenyl)[1,2,5]oxadiazolo[3,4-b]pyrazine-5,6-diamine) is a synthetic mitochondrial uncoupler that functions as a lipophilic weak acid protonophore. Unlike traditional uncouplers such as 2,4-dinitrophenol (DNP), BAM15 demonstrates improved safety profile with reduced off-target effects, particularly avoiding plasma membrane depolarization that characterized earlier generation compounds.
The compound operates by enhancing proton permeability across the inner mitochondrial membrane, dissipating the proton gradient that normally drives ATP synthesis. This uncoupling forces mitochondria to increase substrate oxidation to meet cellular energy demands, resulting in enhanced energy expenditure and metabolic rate. The increased respiration occurs without generating excessive reactive oxygen species or affecting body temperature, distinguishing BAM15 from previous uncoupling agents.
BAM15 demonstrates selective tissue distribution, concentrating primarily in lipid-rich tissues including adipose tissue and liver, with lesser accumulation in skeletal muscle. This preferential targeting underlies its remarkable ability to promote fat loss while preserving lean muscle mass during weight reduction. The compound is orally bioavailable with a half-life of approximately 1.7-3 hours, allowing for practical administration and dosing flexibility.
The mechanism of action involves multiple beneficial pathways beyond simple uncoupling. BAM15 activates AMP-activated protein kinase (AMPK), a master regulator of cellular energy metabolism, leading to enhanced glucose uptake, increased fatty acid oxidation, and improved insulin sensitivity. The compound also activates peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PGC-1α), promoting mitochondrial biogenesis and metabolic adaptation. Additionally, BAM15 inhibits NLRP3 inflammasome activation by suppressing NF-κB nuclear translocation, providing anti-inflammatory effects that contribute to metabolic health improvements.
Unlike appetite suppressants or caloric restriction interventions, BAM15 does not reduce food intake or suppress appetite. Weight loss achieved through BAM15 treatment occurs exclusively through increased energy expenditure and enhanced metabolic efficiency, avoiding the psychological burden and compensatory metabolic adaptations associated with calorie-restricted diets.
Bam15 Structure
Chemical Structure
2D Structure
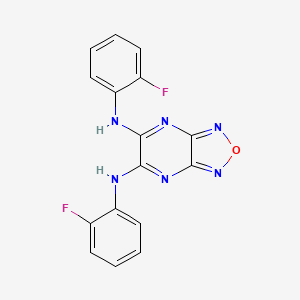
3D Structure

Chemical Properties
| CAS Number | 210302-17-3 |
|---|---|
| Molecular Formula | C16H10F2N6O |
| Molecular Weight | 340.29 g/mol |
| IUPAC Name |
5-N,6-N-bis(2-fluorophenyl)-[1,2,5]oxadiazolo[3,4-b]pyrazine-5,6-diamine
|
| InChIKey | OEGJBRZAJRPPHL-UHFFFAOYSA-N |
Bam15 Research
BAM15 Product Content
Basic Description (700 characters)
BAM15 is a selective mitochondrial uncoupler that enhances metabolic efficiency by disrupting the coupling between electron transport and ATP synthesis in mitochondria. Research demonstrates BAM15 significantly increases energy expenditure, promotes fat loss while preserving lean muscle mass, and improves insulin sensitivity. In animal studies, BAM15 prevented diet-induced obesity, decreased fat mass by 20-30%, reversed insulin resistance, and improved metabolic markers without altering body temperature or food intake. The compound targets lipid-rich tissues, reduces hepatic steatosis, and activates AMPK-mediated fatty acid oxidation. Studies show BAM15 preserves muscle during weight loss, enhances mitochondrial function, and reduces inflammation through NLRP3 inflammasome inhibition.
What is BAM15?
BAM15 (N5,N6-bis(2-Fluorophenyl)[1,2,5]oxadiazolo[3,4-b]pyrazine-5,6-diamine) is a synthetic mitochondrial uncoupler that functions as a lipophilic weak acid protonophore. Unlike traditional uncouplers such as 2,4-dinitrophenol (DNP), BAM15 demonstrates improved safety profile with reduced off-target effects, particularly avoiding plasma membrane depolarization that characterized earlier generation compounds.
The compound operates by enhancing proton permeability across the inner mitochondrial membrane, dissipating the proton gradient that normally drives ATP synthesis. This uncoupling forces mitochondria to increase substrate oxidation to meet cellular energy demands, resulting in enhanced energy expenditure and metabolic rate. The increased respiration occurs without generating excessive reactive oxygen species or affecting body temperature, distinguishing BAM15 from previous uncoupling agents.
BAM15 demonstrates selective tissue distribution, concentrating primarily in lipid-rich tissues including adipose tissue and liver, with lesser accumulation in skeletal muscle. This preferential targeting underlies its remarkable ability to promote fat loss while preserving lean muscle mass during weight reduction. The compound is orally bioavailable with a half-life of approximately 1.7-3 hours, allowing for practical administration and dosing flexibility.
The mechanism of action involves multiple beneficial pathways beyond simple uncoupling. BAM15 activates AMP-activated protein kinase (AMPK), a master regulator of cellular energy metabolism, leading to enhanced glucose uptake, increased fatty acid oxidation, and improved insulin sensitivity. The compound also activates peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PGC-1α), promoting mitochondrial biogenesis and metabolic adaptation. Additionally, BAM15 inhibits NLRP3 inflammasome activation by suppressing NF-κB nuclear translocation, providing anti-inflammatory effects that contribute to metabolic health improvements.
Unlike appetite suppressants or caloric restriction interventions, BAM15 does not reduce food intake or suppress appetite. Weight loss achieved through BAM15 treatment occurs exclusively through increased energy expenditure and enhanced metabolic efficiency, avoiding the psychological burden and compensatory metabolic adaptations associated with calorie-restricted diets.
Research Applications
Fat Loss and Obesity Management
BAM15 demonstrates powerful anti-obesity effects through enhanced mitochondrial uncoupling and increased energy expenditure. Research published in Nature Communications shows that BAM15 treatment decreased body fat mass by 20-30% in diet-induced obese mice without altering food intake, lean body mass, or body temperature. In both prevention and reversal obesity models, mice treated with BAM15 exhibited complete resistance to weight gain or reversal of existing obesity within 3 weeks of treatment.
The compound primarily increases oxygen consumption through fat oxidation rather than increased physical activity. Metabolic cage studies reveal BAM15-treated animals display lower respiratory exchange ratios with elevated plasma non-esterified fatty acid levels, indicating enhanced liberation and oxidation of adipose-derived fatty acids. Direct measurements show BAM15 increases 14C-palmitate oxidation by 54% in liver tissue, demonstrating tissue-specific enhancement of fatty acid metabolism.
BAM15 significantly downregulates key adipogenic transcription factors and lipogenesis genes in white adipose tissue. Gene expression analysis reveals 490-fold downregulation of sterol regulatory element-binding transcription factor 1 (SREBF1), 99-fold reduction in carbohydrate-responsive element-binding protein, and 898-fold decrease in stearoyl-CoA desaturase-1 (SCD1). These molecular changes translate to dramatic reductions in all fat depot weights including gonadal, inguinal, and retroperitoneal white adipose tissue as well as brown adipose tissue.
Sources:
- Alexopoulos SJ, et al. "Mitochondrial uncoupler BAM15 reverses diet-induced obesity and insulin resistance in mice." Nature Communications. 2020;11:2397. https://www.nature.com/articles/s41467-020-16298-2
- Axelrod CL, et al. "BAM15-mediated mitochondrial uncoupling protects against obesity and improves glycemic control." EMBO Molecular Medicine. 2020;12(7):e12088. https://www.embopress.org/doi/10.15252/emmm.202012088
- Qiu J, et al. "BAM15 as a mitochondrial uncoupler: a promising therapeutic agent for diverse diseases." Frontiers in Endocrinology. 2023;14:1252141. https://www.frontiersin.org/journals/endocrinology/articles/10.3389/fendo.2023.1252141/full
Lean Mass Preservation and Muscle Health
BAM15 distinguishes itself from other weight loss interventions through its remarkable ability to preserve and even enhance lean muscle mass during fat loss. Studies demonstrate that while control animals gain both fat and lean mass during high-fat feeding, BAM15-treated animals display reduced fat mass with no change in lean mass compared to baseline. More impressively, when compared to calorie restriction achieving equivalent weight loss, BAM15 treatment results in significantly greater lean mass preservation.
Research on aged obese mice shows BAM15 treatment for 10 weeks increased lean muscle mass by 8%, muscle strength by 40%, and spontaneous physical activity, while simultaneously reducing fat mass by 20%. These improvements in physical function were mediated by reductions in skeletal muscle inflammation (decreased IL-6 and gp130), enhanced mitochondrial function, and improved endoplasmic reticulum homeostasis. Cross-sectional analysis revealed increased muscle fiber area in BAM15-treated animals compared to controls.
The muscle-preserving effect stems from BAM15's tissue distribution pattern and metabolic effects. The compound concentrates in adipose tissue rather than skeletal muscle, coupled with sustained food intake in treated animals. This contrasts sharply with calorie restriction or appetite suppressants like phentermine, which reduce lean body mass through decreased protein intake and energy availability. BAM15 maintains protein synthesis while specifically targeting fat oxidation, allowing simultaneous fat loss and muscle preservation.
In sarcopenic obesity models, mitochondrial uncoupling with BAM15 simultaneously attenuates loss of muscle function and prevents weight gain. The compound enhances skeletal muscle mitophagy and mitochondrial quality control processes, addressing the mitochondrial dysfunction central to age-related muscle decline. Combination studies with semaglutide demonstrate that adding BAM15 preserves and even increases quadriceps weight during weight loss, with percentage fat-free lean mass significantly increased compared to drug treatment alone.
Sources:
- Dantas WS, et al. "Mitochondrial uncoupling attenuates sarcopenic obesity by enhancing skeletal muscle mitophagy and quality control." Journal of Cachexia, Sarcopenia and Muscle. 2022;13(2):1137-1148. https://pubmed.ncbi.nlm.nih.gov/35304976/
- Alpert AJ, et al. "Beneficial effects of simultaneously targeting calorie intake and calorie efficiency in diet-induced obese mice." Clinical Science. 2024;138(4):173-193. https://portlandpress.com/clinsci/article/138/4/173/234024/Beneficial-effects-of-simultaneously-targeting
- Alexopoulos SJ, et al. "Mitochondrial uncoupler BAM15 reverses diet-induced obesity and insulin resistance in mice." Nature Communications. 2020;11:2397. https://pmc.ncbi.nlm.nih.gov/articles/PMC7224297/
Metabolic Health and Insulin Sensitivity
BAM15 demonstrates profound effects on glucose homeostasis and insulin sensitivity across multiple tissue types. Within 3 weeks of treatment, BAM15 completely reversed diet-induced glucose intolerance and hyperinsulinemia in obese mice. Hyperinsulinemic-euglycemic clamp studies—the gold standard for measuring insulin sensitivity—show BAM15 improved whole-body insulin sensitivity to levels equivalent to lean control animals, with normalization of glucose infusion rates and improvements in skeletal muscle glucose clearance.
The compound enhances insulin action through sustained AMPK activation, which promotes GLUT4 expression and glucose uptake independent of insulin stimulation. In vitro studies demonstrate BAM15 improves insulin signaling and stimulates nutrient uptake in muscle cells at concentrations as low as 1-5 μM. Metabolomic analyses reveal BAM15 reduces metabolic pathways associated with obesity and type 2 diabetes, including sphingolipid metabolism, monoacylglycerol metabolism, and dicarboxylate metabolism.
BAM15 treatment significantly lowers fasting plasma glucose and insulin concentrations, improves glucose tolerance test results, and reduces circulating markers of insulin resistance. The compound effectively lowers glucagon secretion and hepatic glucose output by downregulating critical gluconeogenesis enzymes including glucose-6-phosphatase and fructose-1,6-bisphosphatase, alleviating hyperglycemia while preserving pancreatic alpha cell mass.
Comparative studies demonstrate BAM15's superior metabolic effects. In female db/db mice—a severe metabolic disease model—BAM15 completely restored glucose tolerance to levels comparable to semaglutide and rosiglitazone, while achieving superior reductions in body weight and liver fat. These improvements occur through enhanced metabolic flexibility—the capacity to switch between carbohydrate and fat oxidation—making BAM15 particularly valuable for combating metabolic syndrome.
Sources:
- Alexopoulos SJ, et al. "Mitochondrial uncoupler BAM15 reverses diet-induced obesity and insulin resistance in mice." Nature Communications. 2020;11:2397. https://www.nature.com/articles/s41467-020-16298-2
- Axelrod CL, et al. "BAM15-mediated mitochondrial uncoupling protects against obesity and improves glycemic control." EMBO Molecular Medicine. 2020;12(7):e12088. https://www.embopress.org/doi/10.15252/emmm.202012088
- Donahoo AK, et al. "Head-to-head comparison of BAM15, semaglutide, rosiglitazone, NEN, and calorie restriction on metabolic physiology in female db/db mice." Biochimica et Biophysica Acta. 2024;1870(1):167331. https://www.sciencedirect.com/science/article/pii/S0925443923002740
Liver Health and NAFLD Protection
BAM15 shows significant therapeutic potential for nonalcoholic fatty liver disease (NAFLD) and nonalcoholic steatohepatitis (NASH) through multiple protective mechanisms. Studies demonstrate BAM15 treatment dramatically reduces hepatic steatosis, decreases liver triglyceride levels, and improves liver enzyme markers in multiple animal models of metabolic liver disease. In db/db mice fed high-fat diets, BAM15 reduced liver triglycerides and serum ALT and AST levels while alleviating oxidative stress and mitochondrial dysfunction.
The compound's liver-protective effects stem from enhanced hepatic fatty acid oxidation and reduced lipogenesis. BAM15 increases 14C-palmitate oxidation in liver tissue ex vivo and decreases hepatic triglyceride and non-esterified fatty acid accumulation. Histological analyses reveal marked reductions in lipid droplet accumulation in hepatocytes, with liver weight normalized in treated animals compared to obese controls.
In NASH models, BAM15 derivatives reduced liver triglyceride levels by 20-30% and showed improvements in fibrosis, inflammation, and plasma ALT levels. The compound's effects on NAFLD Activity Score encompassed improvements in all three parameters: steatosis, inflammation, and hepatocellular ballooning. Drug-albumin nanocomposites of BAM15 demonstrated enhanced liver-targeting capacity, effectively alleviating hepatic steatosis and improving therapeutic efficacy compared to free BAM15.
BAM15's mechanism in liver protection involves AMPK activation in hepatic tissue, leading to suppression of lipogenic gene expression. The compound reduces expression of key fat synthesis enzymes including fatty acid synthase (FASN), stearoyl-CoA desaturase-1 (SCD1), and sterol regulatory element-binding transcription factor 1 (SREBF1). Additionally, BAM15 mitigates the NLRP3 inflammasome activation that contributes to NAFLD pathogenesis, reducing inflammatory cytokine production and hepatic inflammation.
Sources:
- Qiu J, et al. "BAM15 as a mitochondrial uncoupler: a promising therapeutic agent for diverse diseases." Frontiers in Endocrinology. 2023;14:1252141. https://pmc.ncbi.nlm.nih.gov/articles/PMC10600450/
- Childress ES, et al. "[1,2,5]Oxadiazolo[3,4-b]pyrazine-5,6-diamine Derivatives as Mitochondrial Uncouplers for the Potential Treatment of Nonalcoholic Steatohepatitis." Journal of Medicinal Chemistry. 2020;63(5):2511-2526. https://pubmed.ncbi.nlm.nih.gov/32017849/
- Jing W, et al. "Self-assembly drug-albumin nanocomposites for nonalcoholic fatty liver disease treatment." International Journal of Biological Macromolecules. 2022;215:625-637. https://www.sciencedirect.com/science/article/abs/pii/S0141813022013800
Cardiovascular Protection and Atherosclerosis Prevention
BAM15 demonstrates significant cardiovascular protective effects through multiple mechanisms including reduced inflammation, improved lipid profiles, and enhanced endothelial function. Research on atherosclerosis models shows oral administration of BAM15 suppressed atherosclerotic plaque formation in Western diet-fed ApoE-/- mice, with significant improvements in hyperlipidemia markers. Histological analyses revealed reduced lipid deposition in aortic tissue and decreased inflammatory cytokine levels including IL-1β and IL-18.
The compound protects endothelial cells from hyperglycemia-induced injury and apoptosis. Studies demonstrate BAM15 enhances fatty acid oxidation, reduces reactive oxygen species production, and improves endothelial cell survival under diabetic conditions. Low-dose BAM15 treatment outperformed insulin in promoting endothelial cell viability, while also improving mitochondrial complex I and IV activity and increasing ATP production in endothelial cells. Gene expression analysis revealed BAM15 restored expression of fatty acid oxidation genes suppressed by hyperglycemia.
BAM15's cardiovascular benefits extend to direct anti-atherosclerotic mechanisms. The compound reduces atherosclerotic plaque formation, lipid deposition, and inflammation while diminishing mitochondrial ROS expression and oxidized mitochondrial DNA content. In both in vivo and in vitro experiments, BAM15 markedly inhibited activation of the NLRP3 inflammasome in endothelial cells, leading to reduced pyroptosis—a form of inflammatory cell death implicated in plaque instability. The compound suppresses NF-κB nuclear translocation and inhibits expression of pro-inflammatory markers PTX3, which plays roles in vascular injury and atherosclerosis progression.
BAM15 exhibits biphasic effects on STAT3 activity—a critical signaling molecule in cardiac protection. At low doses, BAM15 activates STAT3 through the mitochondrial ROS/JAK/STAT3 pathway, inducing Tyr705 phosphorylation that results in reduced cardiac injury and increased ATP production. This cardioprotective signaling makes BAM15 a promising candidate for preventing ischemic injury. Additionally, the compound activates AMPK in vascular smooth muscle cells with higher potency than metformin or AICAR, contributing to improved vascular function and reduced atherosclerotic burden.
Sources:
- Li X, et al. "BAM15 inhibits endothelial pyroptosis via the NLRP3/ASC/caspase-1 pathway to alleviate atherosclerosis." Journal of Inflammation Research. 2025;18:2437-2453. https://www.sciencedirect.com/science/article/abs/pii/S0021915025001248
- Younes NA, et al. "Targeting Mitochondrial Dysfunction to Prevent Endothelial Dysfunction and Atherosclerosis in Diabetes: Focus on the Novel Uncoupler BAM15." International Journal of Molecular Sciences. 2025;26(10):4603. https://www.mdpi.com/1422-0067/26/10/4603
- Wu Y, et al. "Combining RNA-seq, molecular docking and experimental verification to explore the mechanism of BAM15 as a potential drug for atherosclerosis." Scientific Reports. 2025;15:2562. https://www.nature.com/articles/s41598-025-98209-3
Anti-Inflammatory and Immunomodulatory Effects
BAM15 exerts potent anti-inflammatory effects through inhibition of the NLRP3 inflammasome, a multiprotein complex implicated in numerous inflammatory diseases. Research demonstrates BAM15 specifically targets the initiation step of NLRP3 inflammasome activation by suppressing NF-κB translocation to the nucleus, thereby inhibiting expression of NLRP3 and pro-inflammatory cytokine IL-1β. The compound also increases intracellular calcium concentration and inhibits IκBα phosphorylation, providing additional mechanisms for blocking inflammatory signaling cascades.
Studies in macrophages show BAM15 reduces production of inflammatory cytokines including IL-6, TNF-α, IL-1β, and IL-18. These reductions occur both at the transcriptional level through suppressed gene expression and at the post-translational level through inhibited inflammasome-mediated processing of pro-inflammatory cytokines. In vivo studies demonstrate BAM15 treatment reduces circulating levels of leptin, GDF15, FGF21, and other inflammatory markers associated with obesity and metabolic dysfunction.
The anti-inflammatory effects extend to tissue-specific improvements. In skeletal muscle of aged obese mice, BAM15 treatment reduced IL-6 and gp130 expression, markers of chronic low-grade inflammation that contribute to muscle atrophy and metabolic dysfunction. In liver tissue, BAM15 decreased NLRP3 inflammasome activation and reduced hepatic inflammation scores in NASH models. The compound's ability to modulate macrophage polarization contributes to resolution of inflammatory processes in multiple tissues.
BAM15 demonstrates therapeutic potential in sepsis through anti-inflammatory mechanisms. In cecal ligation and puncture models, BAM15 treatment reduced mortality even when administered 12 hours after sepsis induction. The compound reduced mitochondrial DNA release—a damage-associated molecular pattern that triggers inflammatory responses—and mitigated kidney damage and splenic apoptosis. BAM15 treatment lowered circulating inflammatory cytokines and reduced neutrophil infiltration, demonstrating broad immunomodulatory effects that extend beyond simple metabolic improvements.
Sources:
- Hu C, et al. "Chemical mitochondrial uncouplers share common inhibitory effect on NLRP3 inflammasome activation through inhibiting NFκB nuclear translocation." Toxicology and Applied Pharmacology. 2021;414:115426. https://pubmed.ncbi.nlm.nih.gov/33508337/
- Qiu J, et al. "BAM15 as a mitochondrial uncoupler: a promising therapeutic agent for diverse diseases." Frontiers in Endocrinology. 2023;14:1252141. https://www.frontiersin.org/journals/endocrinology/articles/10.3389/fendo.2023.1252141/full
- Tsuji S, et al. "BAM15 treats mouse sepsis and kidney injury, linking mortality, mitochondrial DNA, tubule damage, and neutrophils." Journal of Clinical Investigation. 2023;133(7):e152401. https://www.jci.org/articles/view/152401
Age-Related Muscle Function and Sarcopenia
BAM15 demonstrates significant potential for addressing age-related muscle decline and sarcopenia through enhancement of mitochondrial function and metabolic efficiency. In 24-month-old mice (equivalent to elderly humans), 8 weeks of BAM15 supplementation improved skeletal muscle contractile function and mitochondrial respiration without altering age-related loss in muscle mass. The compound preserved extensor digitorum longus contractile function that typically declines with age, demonstrating functional preservation despite chronological aging.
Mitochondrial respiration analyses in aged muscle revealed BAM15 supplementation protected against age-related declines in Complex I and Complex I+II substrate-dependent respiration. The compound increased ADP-dependent respiration and maintained Complex I leak respiration, suggestive of mild beneficial uncoupling that improves mitochondrial efficiency. These improvements in mitochondrial function translated to preserved muscle quality and contractile capacity, with muscle generating greater force per unit mass compared to untreated aged controls.
Research on sarcopenic obesity shows particularly striking results. In 80-week-old obese mice treated with BAM15 for 10 weeks, lean muscle mass increased by 8%, grip strength improved by 40%, and spontaneous physical activity increased significantly. Muscle fiber cross-sectional area was significantly greater in BAM15-treated animals, indicating both quantitative and qualitative improvements in muscle tissue. These functional improvements occurred concomitantly with 20% reduction in fat mass, demonstrating BAM15's unique ability to simultaneously address both components of sarcopenic obesity.
The mechanisms underlying muscle preservation and enhancement involve multiple pathways. BAM15 reduces skeletal muscle inflammation by decreasing IL-6 and gp130 expression, key mediators of age-related muscle atrophy. The compound enhances mitochondrial quality control through improved mitophagy and endoplasmic reticulum homeostasis, addressing fundamental causes of age-related mitochondrial dysfunction. Additionally, BAM15 treatment maintains oxidative stress balance and redox homeostasis, preventing the accumulation of damaged mitochondria and oxidized proteins that impair muscle function during aging.
Sources:
- Holloway TM, et al. "Mitochondrial uncoupler BAM15 improves skeletal muscle function and mitochondrial respiration in Sarcopenia." bioRxiv. 2025. https://www.biorxiv.org/content/10.1101/2025.10.30.685477v1.full
- Dantas WS, et al. "Mitochondrial uncoupling attenuates sarcopenic obesity by enhancing skeletal muscle mitophagy and quality control." Journal of Cachexia, Sarcopenia and Muscle. 2022;13(2):1137-1148. https://pubmed.ncbi.nlm.nih.gov/35304976/
- Axelrod CL, et al. "BAM15-mediated mitochondrial uncoupling protects against obesity and improves glycemic control." EMBO Molecular Medicine. 2020;12(7):e12088. https://pmc.ncbi.nlm.nih.gov/articles/PMC7338798/


