⊗This PRODUCT IS INTENDED AS A RESEARCH CHEMICAL ONLY. This designation allows the use of research chemicals strictly for in vitro testing and laboratory experimentation only. All product information available on this website is for educational purposes only. Bodily introduction of any kind into humans or animals is strictly prohibited by law. This product should only be handled by licensed, qualified professionals. This product is not a drug, food, or cosmetic and may not be misbranded, misused or mislabeled as a drug, food, or cosmetic.
⊗This PRODUCT IS INTENDED AS A RESEARCH CHEMICAL ONLY. This designation allows the use of research chemicals strictly for in vitro testing and laboratory experimentation only. All product information available on this website is for educational purposes only. Bodily introduction of any kind into humans or animals is strictly prohibited by law. This product should only be handled by licensed, qualified professionals. This product is not a drug, food, or cosmetic and may not be misbranded, misused or mislabeled as a drug, food, or cosmetic.
GHRP-6 5mg
Peptides
What is GHRP-6?
GHRP-6 (Growth Hormone-Releasing Peptide-6) is a synthetic hexapeptide (His-DTrp-Ala-Trp-DPhe-Lys-NH2) developed as a growth hormone secretagogue that stimulates endogenous growth hormone release through mechanisms distinct from growth hormone-releasing hormone (GHRH). Originally synthesized as a derivative of met-enkephalin, GHRP-6 was the first in its class to specifically elicit dose-related growth hormone release both in vitro and in vivo.
The peptide functions by binding to the growth hormone secretagogue receptor (GHS-R1a), also known as the ghrelin receptor, located in the pituitary gland and hypothalamus. This receptor activation initiates a signaling cascade involving protein kinase C (PKC) and calcium mobilization, leading to robust growth hormone secretion. Unlike GHRH, which operates through cyclic AMP pathways, GHRP-6 stimulates phosphatidylinositol (PI) turnover as its primary second messenger system. When administered with GHRH, GHRP-6 produces a synergistic effect that dramatically amplifies growth hormone release beyond the sum of individual responses.
GHRP-6 also binds to CD36, a scavenger receptor abundantly expressed in cardiovascular tissue, skeletal muscle, and cutaneous wounds. This dual receptor binding mechanism accounts for GHRP-6's diverse biological effects extending far beyond growth hormone stimulation. The peptide's interaction with CD36 mediates crucial cytoprotective, anti-inflammatory, and anti-fibrotic effects independent of the growth hormone axis.
Following administration, GHRP-6 triggers a substantial increase in circulating growth hormone levels, with peak responses occurring within 15-30 minutes. This growth hormone surge subsequently stimulates hepatic production of insulin-like growth factor-1 (IGF-1), which remains elevated for prolonged periods and mediates many of GHRP-6's anabolic and regenerative effects. IGF-1 plays essential roles in protein synthesis, satellite cell activation, collagen production, and tissue repair processes throughout the body.
Research demonstrates GHRP-6 is effective through multiple administration routes including intravenous, subcutaneous, intranasal, and oral delivery. The peptide exhibits remarkable stability, low molecular weight, and an established safety profile in human clinical trials. Importantly, GHRP-6's growth hormone-releasing activity has been shown to require endogenous GHRH for maximal effect, indicating the peptide works synergistically with the body's natural growth hormone regulatory system rather than bypassing it entirely.
GHRP-6 Structure
Chemical Structure
2D Structure
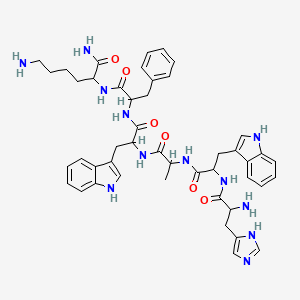
3D Structure

Chemical Properties
| Molecular Formula | C46H56N12O6 |
|---|---|
| Molecular Weight | 873.0 g/mol |
| IUPAC Name |
6-amino-2-[[2-[[2-[2-[[2-[[2-amino-3-(1H-imidazol-5-yl)propanoyl]amino]-3-(1H-indol-3-yl)propanoyl]amino]propanoylamino]-3-(1H-indol-3-yl)propanoyl]amino]-3-phenylpropanoyl]amino]hexanamide
|
| InChIKey | WZHKXNSOCOQYQX-UHFFFAOYSA-N |
GHRP-6 Research
Growth Hormone and IGF-1 Secretion
GHRP-6 demonstrates potent growth hormone-releasing activity across multiple species and clinical populations. Research in healthy adult males shows GHRP-6 administration (1 μg/kg intravenous) produces peak growth hormone levels of 33.8 ± 4.8 μg/L, significantly exceeding baseline concentrations. Studies comparing GHRP-6 to maximally effective doses of GHRH reveal GHRP-6 is more efficacious, with sustained growth hormone responses that translate to prolonged IGF-1 elevation.
Clinical investigations demonstrate GHRP-6's mechanism requires endogenous GHRH for maximal growth hormone stimulation. When GHRH antagonists are administered, the growth hormone response to GHRP-6 is reduced by approximately 80-85%, confirming the peptide amplifies rather than replaces natural growth hormone secretory patterns. This synergistic relationship makes GHRP-6 particularly valuable for maintaining physiologic pulsatile growth hormone release rather than producing sustained non-physiologic elevation.
Research in hypogonadal men receiving combined GHRP-6, GHRP-2, and sermorelin therapy documents significant increases in serum IGF-1 levels at 90, 180, and 270 days of treatment, with improvements in lean body mass and metabolic parameters. The peptide's ability to stimulate IGF-1 production extends to peripheral tissues, with studies showing increased IGF-1 mRNA expression in skeletal muscle, cardiac muscle, brain, and hypothalamus following GHRP-6 administration.
GHRP-6's growth hormone-releasing activity is influenced by metabolic factors. When administered simultaneously with insulin, GHRP-6's growth hormone response is significantly enhanced. However, consumption of carbohydrates or dietary fats around the administration window substantially blunts growth hormone release, suggesting optimal timing relative to meals is important for maximizing therapeutic effects.
Sources:
- Pandya N, et al. "Growth hormone (GH)-releasing peptide-6 requires endogenous hypothalamic GH-releasing hormone for maximal GH stimulation." Journal of Clinical Endocrinology & Metabolism. 1998;83(4):1186-1189. https://pubmed.ncbi.nlm.nih.gov/9543138/
- Adams EF, et al. "Growth hormone releasing peptide (GHRP-6) stimulates phosphatidylinositol (PI) turnover in human pituitary somatotroph cells." Journal of Endocrinology. 1995;147(3):417-423. https://pubmed.ncbi.nlm.nih.gov/7772238/
- Sigalos JT, et al. "Beyond the androgen receptor: the role of growth hormone secretagogues in the modern management of body composition in hypogonadal males." Therapeutic Advances in Urology. 2020;12:1-14. https://pmc.ncbi.nlm.nih.gov/articles/PMC7108996/
Muscle Growth and Body Composition
GHRP-6 promotes muscle development through multiple mechanisms involving growth hormone, IGF-1, and direct cellular effects. Research demonstrates GHRP-6-biotin conjugate treatment significantly increases IGF-1 expression in myoblasts, stimulating myogenic differentiation and collagen type I synthesis essential for muscle regeneration. The peptide upregulates myogenic regulatory factors including MyoD and Myf5, while promoting satellite cell activation critical for muscle hypertrophy.
Studies in normal mice show GHRP-6 administration produces significant improvements in body composition, muscle growth, and glucose metabolism. The peptide increases lean body mass through enhanced protein synthesis while simultaneously promoting lipolysis and fat utilization. Research indicates GHRP-6 activates anabolic pathways including IGF-1/AKT1 and mTOR signaling, which drive muscle protein synthesis and cellular growth. These effects occur independently of exogenous growth hormone administration, demonstrating GHRP-6's direct myotropic properties.
Animal studies reveal GHRP-6 increases skeletal muscle IGF-1 mRNA and protein levels following systemic administration. The sustained elevation of IGF-1 promotes anabolic processes while inhibiting catabolism, protecting muscle tissue from degradation during metabolic stress. Research demonstrates GHRP-6 increases desmin expression, a muscle-specific intermediate filament protein critical for maintaining structural and mechanical integrity of contractile apparatus.
GHRP-6 exhibits appetite-stimulating effects through ghrelin receptor activation, which can support increased caloric intake and nutrient availability for muscle growth. This orexigenic effect distinguishes GHRP-6 from other growth hormone secretagogues and can be strategically utilized during periods requiring enhanced nutritional support for tissue building and recovery.
Sources:
- Kim SH, et al. "Growth hormone-releasing peptide-biotin conjugate stimulates myocytes differentiation through insulin-like growth factor-1 and collagen type I." Molecular and Cellular Endocrinology. 2015;418:110-119. https://pmc.ncbi.nlm.nih.gov/articles/PMC4641233/
- Berlanga-Acosta J, et al. "Synthetic Growth Hormone-Releasing Peptides (GHRPs): A Historical Appraisal of the Evidences Supporting Their Cytoprotective Effects." Clinical Medicine Insights: Cardiology. 2017;11:1-23. https://pmc.ncbi.nlm.nih.gov/articles/PMC5392015/
Wound Healing and Tissue Repair
GHRP-6 demonstrates remarkable wound healing acceleration and anti-fibrotic properties through CD36 receptor activation. Research in full-thickness wound models shows GHRP-6 topical application (400 μg/mL) significantly accelerates wound closure beginning within 24 hours of initial treatment, with effects remaining stable through 96-hour observation periods. The peptide achieves this through multiple coordinated mechanisms including reduced inflammatory infiltration, enhanced collagen organization, and modulation of fibrogenic cytokine expression.
Studies reveal GHRP-6 attenuates immunoinflammatory mediators and reduces expression of pro-fibrotic genes including transforming growth factor-β1 (TGF-β1), connective tissue growth factor (CTGF), and tumor necrosis factor converting enzyme (ADAM17). Molecular analyses demonstrate GHRP-6 treatment decreases TGF-β1 transcription by up to 60% while simultaneously activating PPARγ, a key regulator that opposes fibrogenic processes. This anti-fibrotic mechanism proves particularly valuable in preventing pathological scarring.
In hypertrophic scar models, GHRP-6 prevents the onset of exuberant scars in over 90% of treated wounds, dramatically reducing scar elevation index and sub-epidermal fibrotic core accumulation. The peptide demonstrates superior outcomes compared to triamcinolone acetonide, the first-line pharmaceutical treatment, without associated adverse effects. Histological analyses reveal GHRP-6-treated wounds exhibit organized extracellular matrix with horizontally distributed collagen strands rather than the dense irregular accumulation characteristic of hypertrophic scarring.
Research indicates GHRP-6's wound healing effects involve enhanced collagen synthesis with improved organization rather than excessive deposition. The peptide increases expression of matrix metalloproteinase-13 (MMP-13), promoting appropriate matrix remodeling during healing phases. Proteomics studies identify GHRP-6 modulates proteins involved in lipid metabolism, cytoskeleton arrangements, ECM-cell interactions, and epidermal cell differentiation within one hour of application, demonstrating rapid mechanistic engagement with healing processes.
Sources:
- Mendoza-Marí Y, et al. "Growth Hormone-Releasing Peptide 6 Enhances the Healing Process and Improves the Esthetic Outcome of the Wounds." Plastic Surgery International. 2016;2016:4361702. https://pmc.ncbi.nlm.nih.gov/articles/PMC4854984/
- Fernández-Mayola M, et al. "Growth hormone-releasing peptide 6 prevents cutaneous hypertrophic scarring: early mechanistic data from a proteome study." International Wound Journal. 2018;15(4):568-579. https://pmc.ncbi.nlm.nih.gov/articles/PMC7949743/
- Berlanga-Acosta J, et al. "GHRP-6, a novel candidate for prevention and treatment of fibrotic disorders." Biotecnología Aplicada. 2017;34(3):301-310. http://scielo.sld.cu/scielo.php?script=sci_arttext&pid=S1027-28522017000200006
Cardioprotection and Cardiovascular Health
GHRP-6 exhibits potent cardioprotective effects through multiple mechanisms including antioxidant activity, anti-inflammatory properties, and direct myocardial functional support. Research in acute myocardial infarction models demonstrates GHRP-6 administration (400 μg/kg) reduces infarct mass by 78% and infarct thickness by 50% compared to saline controls. Over 50% of GHRP-6-treated animals show no pathological Q-waves on ECG, indicating prevention of transmural myocardial damage typically associated with severe infarction.
Studies reveal GHRP-6 prevents myocardial injury through reduction of reactive oxygen species and preservation of antioxidant defense systems. Oxidative stress markers including malondialdehyde, protein carbonyls, and lipid peroxides are significantly reduced in GHRP-6-treated subjects, while antioxidant enzymes such as superoxide dismutase and glutathione peroxidase remain elevated. Histopathological analyses confirm GHRP-6 preserves myocardial fiber integrity adjacent to infarct zones, with minimal myofibrolysis compared to extensive damage in untreated controls.
GHRP-6 demonstrates positive inotropic effects independent of heart rate changes. Echocardiographic studies show GHRP-6 administration increases left ventricular ejection fraction in a dose-dependent manner (100-400 μg/kg), with effects sustained even in subjects chronically treated with beta-blockers. This indicates GHRP-6's cardiac functional improvements do not depend on beta-adrenergic stimulation, suggesting alternative receptor-mediated mechanisms through GHS-R1a and CD36 pathways.
Research in doxorubicin-induced cardiomyopathy models reveals GHRP-6 prevents development of dilated cardiomyopathy and heart failure. GHRP-6 treatment preserves left ventricular systolic function, prevents ventricular dilation, and maintains myocardial fiber structure. The peptide upregulates anti-apoptotic gene Bcl-2 while sustaining mitochondrial integrity, protecting cardiomyocytes from drug-induced cytotoxicity. Importantly, GHRP-6 exhibits broad extracardiac cytoprotective effects, reducing parenchymal necrosis and fibrosis in liver, kidneys, and lungs exposed to toxic insults.
Clinical safety studies in healthy human volunteers confirm GHRP-6 intravenous administration up to 400 μg/kg is well-tolerated with no pharmacological interactions with cardiovascular medications including beta-blockers, supporting its potential therapeutic application in cardiac patients.
Sources:
- Berlanga J, et al. "Growth-hormone-releasing peptide 6 (GHRP6) prevents oxidant cytotoxicity and reduces myocardial necrosis in a model of acute myocardial infarction." Clinical Science. 2007;112(4):241-250. https://pubmed.ncbi.nlm.nih.gov/16989643/
- Valiente J, et al. "Cardiotropic effect of GHRP-6: in vivo characterization by echocardiography." Biotecnología Aplicada. 2013;30(4):285-289. http://scielo.sld.cu/pdf/bta/v30n4/bta05413.pdf
- Puldón-Seguí G, et al. "Growth hormone releasing peptide-6 (GHRP-6) prevents doxorubicin-induced myocardial and extra-myocardial damages by activating prosurvival mechanisms." Frontiers in Pharmacology. 2024;15:1402138. https://www.frontiersin.org/journals/pharmacology/articles/10.3389/fphar.2024.1402138/full
- Garcia del Barco D, et al. "Clinical safety of Growth Hormone-Releasing Peptide 6 (GHRP-6) in healthy volunteers." Biotecnología Aplicada. 2014;31(3):244-248. https://www.medigraphic.com/cgi-bin/new/resumenI.cgi?IDARTICULO=50887
Bone Health and Mineralization
GHRP-6 significantly enhances bone mineral density and promotes bone formation through growth hormone and IGF-1-mediated mechanisms. Research in adult female rats demonstrates continuous GHRP-6 administration (0.5 mg/kg/day for 12 weeks) increases total bone mineral content in tibial and vertebral sites as measured by dual X-ray absorptiometry. While total BMC corrected for body weight remains unchanged, tibial area bone mineral density increases significantly, indicating genuine bone strengthening effects.
Studies using peripheral quantitative computed tomography reveal GHRP-6 increases cortical bone mineral content through enhanced cross-sectional bone area expansion. Femur and vertebral volumes increase substantially, with elevated ash weight confirming true mineralization rather than mere structural changes. These effects mirror those achieved with growth hormone administration, demonstrating GHRP-6's efficacy in promoting skeletal health through endogenous growth hormone stimulation.
Research in ovariectomy-induced osteoporosis models shows GHRP-6 proves equally effective as estrogen replacement therapy in preventing bone loss. GHRP-6 treatment increases serum alkaline phosphatase levels (a marker of bone formation) without elevating acid phosphatase (a marker of bone resorption), indicating a favorable shift toward bone anabolism. Histomorphometric evaluation reveals GHRP-6 preserves trabecular bone volume and architecture that typically deteriorates following ovariectomy.
The mechanism underlying GHRP-6's bone-protective effects involves both systemic growth hormone/IGF-1 elevation and potential direct effects on bone cells. Growth hormone and IGF-1 stimulate osteoblast proliferation, increase collagen synthesis, and enhance bone matrix deposition. Studies demonstrate growth hormone deficiency correlates with reduced bone mineral density and increased fracture risk, while growth hormone replacement reverses these deficits. GHRP-6's ability to restore physiologic growth hormone pulsatility makes it particularly suited for addressing age-related declines in bone mass associated with diminished growth hormone secretion.
Sources:
- Svensson J, et al. "The GH secretagogues ipamorelin and GH-releasing peptide-6 increase bone mineral content in adult female rats." Journal of Endocrinology. 2000;165(3):569-577. https://pubmed.ncbi.nlm.nih.gov/10828840/
- Ibrahim I, et al. "Growth Hormone Releasing Hexapeptide-6 (GHRP-6): Possible Protective Role against Experimentally-Induced Osteoporosis in Female Albino Rats." Bulletin of Egyptian Society for Physiological Sciences. 2012;32(1):77-92. https://besps.journals.ekb.eg/article_35506.html
- Yakar S, et al. "Skeletal Effects of Growth Hormone and Insulin-like Growth Factor-I Therapy." Growth Hormone & IGF Research. 2016;27:28-42. https://pmc.ncbi.nlm.nih.gov/articles/PMC4808510/


