⊗This PRODUCT IS INTENDED AS A RESEARCH CHEMICAL ONLY. This designation allows the use of research chemicals strictly for in vitro testing and laboratory experimentation only. All product information available on this website is for educational purposes only. Bodily introduction of any kind into humans or animals is strictly prohibited by law. This product should only be handled by licensed, qualified professionals. This product is not a drug, food, or cosmetic and may not be misbranded, misused or mislabeled as a drug, food, or cosmetic.
⊗This PRODUCT IS INTENDED AS A RESEARCH CHEMICAL ONLY. This designation allows the use of research chemicals strictly for in vitro testing and laboratory experimentation only. All product information available on this website is for educational purposes only. Bodily introduction of any kind into humans or animals is strictly prohibited by law. This product should only be handled by licensed, qualified professionals. This product is not a drug, food, or cosmetic and may not be misbranded, misused or mislabeled as a drug, food, or cosmetic.
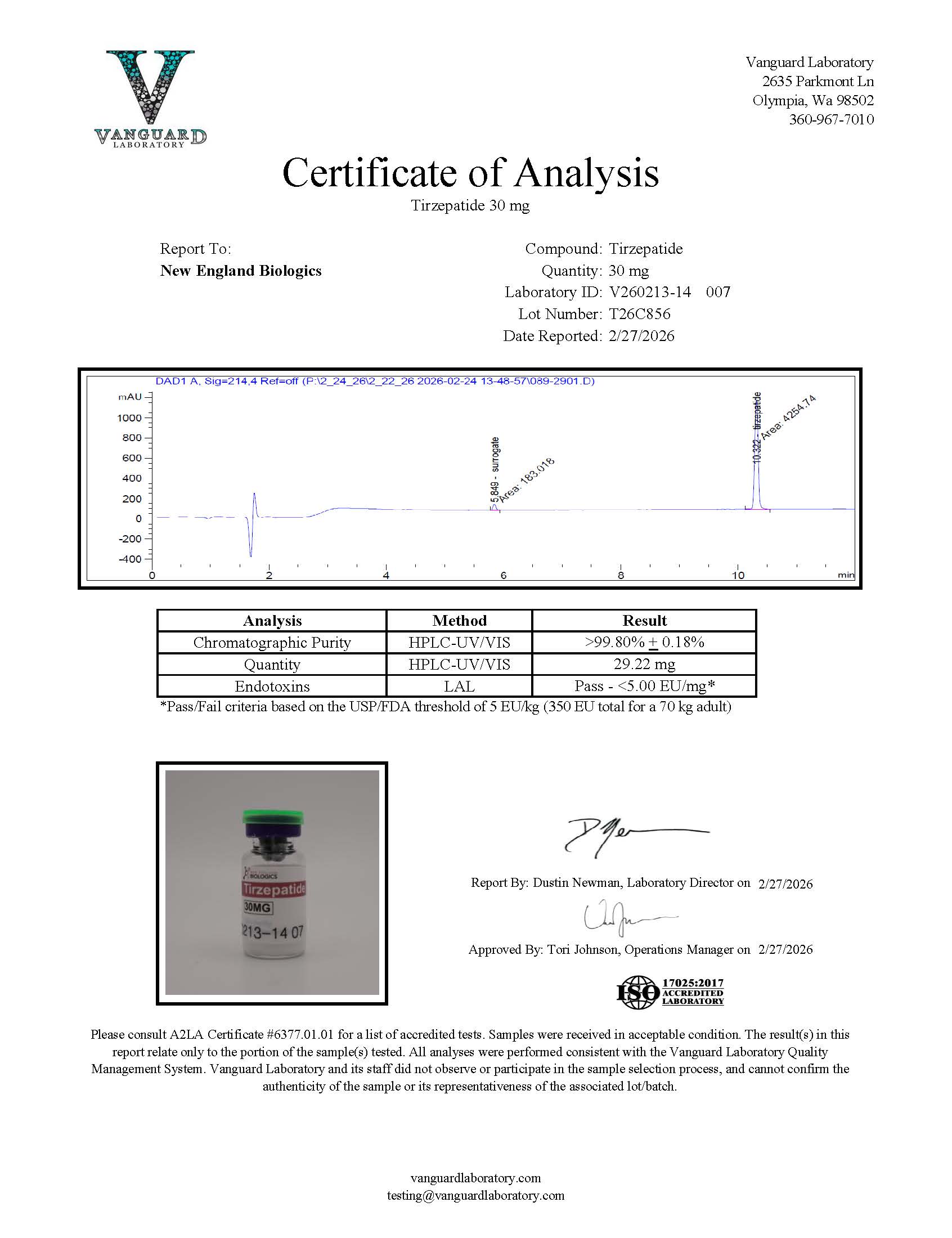
Tirzepatide 30mg
Chemical Properties
| CAS Number | 2023788-19-2 |
|---|---|
| PubChem CID | 156588324 |
| Synonyms | Tirzepatide, P1206, LY3298176, Dual GIP and GLP-1 receptor agonist (GIP/GLP-1RA) |
Peptides
What is Tirzepatide?
Tirzepatide (30 mg) is a synthetic peptide that acts as a dual agonist of the glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptors. It activates both pathways to study coordinated incretin signaling and its effects on endocrine regulation, particularly in areas such as energy balance, regulation of adipose tissue distribution, and metabolic function. New England Biologics™ supplies 30mg tirzepatide produced through solid-phase peptide synthesis (SPPS) and purified by high-performance liquid chromatography (HPLC), consistently delivering purities above 99.9%. With mass spectrometry verification for identity and purity with Certificates of Analysis available, you can buy Tirzepatide 30mg for research use with confidence.
Note: Tirzepatide is supplied strictly for laboratory research use only.
Tirzepatide is a synthetic peptide belonging to the class of dual incretin receptor agonists, designed to interact with both the glucose-dependent insulinotropic polypeptide (GIP) receptor and the glucagon-like peptide-1 (GLP-1) receptor [1]. These receptors are part of the incretin signaling system, a hormone network involved in regulating metabolic communication between organs such as the pancreas, gastrointestinal tract, and brain.
Structurally, tirzepatide is derived primarily from the sequence of the endogenous incretin hormone GIP, with several targeted amino acid substitutions incorporated to enhance receptor interaction and structural stability within experimental systems. These modifications allow the peptide to function as a controlled agonist capable of activating two related incretin receptor pathways in laboratory models.
The molecule also includes a lipid side chain attached through a linker region. This structural feature enables reversible binding to serum albumin, which influences peptide persistence and stability during biochemical and cellular experiments. Lipidation strategies of this type are commonly used in peptide engineering to modify pharmacokinetic and structural characteristics in experimental compounds.
Within laboratory research settings, tirzepatide is studied as a pathway-modulating peptide that activates both incretin receptor systems simultaneously. Activation of GLP-1 and GIP receptors can initiate intracellular signaling cascades that include cyclic AMP generation and other downstream endocrine signaling processes. Because these receptors participate in interconnected metabolic pathways, dual receptor agonists such as tirzepatide allow researchers to investigate receptor cross-talk, signaling bias, and multi-pathway modulation in controlled experimental environments [2].
These structural and functional characteristics make tirzepatide a useful molecular tool for investigating incretin biology, receptor pharmacology, and endocrine signaling mechanisms in biochemical assays and preclinical research systems.
Tirzepatide supplied by New England Biologics is produced using controlled solid-phase peptide synthesis and purified through high-performance liquid chromatography to achieve high levels of chemical purity. Analytical verification procedures confirm molecular identity, purity, and physicochemical stability, supporting consistent performance across laboratory research applications.
Chemical Identity
Tirzepatide consists of a 39–amino acid peptide backbone derived primarily from the sequence of the endogenous incretin hormone glucose-dependent insulinotropic polypeptide. Several targeted amino acid substitutions are incorporated to improve receptor interaction and structural stability in experimental systems.
The peptide also contains a lipid side-chain conjugated through a linker region that facilitates reversible association with serum albumin. This structural modification influences peptide persistence during biochemical assays and contributes to the molecule's ability to function as a dual agonist of both GIP and GLP-1 receptor systems in experimental models.
These molecular features make tirzepatide a useful research compound for investigating coordinated incretin signaling, receptor pharmacology, and endocrine pathway interactions within laboratory settings.
Tirzepatide Research Applications
Tirzepatide is widely studied in laboratory research as a molecular tool for investigating incretin signaling and the metabolic pathways influenced by nutrient-responsive hormones. The compound functions as a dual agonist of the glucose-dependent insulinotropic polypeptide (GIP) receptor and the glucagon-like peptide-1 (GLP-1) receptor, two signaling systems that play central roles in endocrine regulation of metabolism.
Because tirzepatide activates both receptor pathways simultaneously, researchers can use it to explore how coordinated incretin signaling influences intracellular signaling networks, hormone-mediated communication between tissues, and metabolic regulation in controlled experimental systems. Studies involving tirzepatide typically use biochemical assays, cultured cell systems, and preclinical animal models to evaluate how dual receptor activation affects signaling processes across multiple metabolic tissues.
Energy Balance and Metabolic Signaling
One major research area for tirzepatide focuses on how incretin receptor activation influences cellular processes associated with energy metabolism. Experimental investigations frequently examine signaling pathways that regulate nutrient processing, energy storage, and substrate utilization within metabolically active tissues.
Activation of GIP and GLP-1 receptors can initiate intracellular cyclic AMP signaling cascades and downstream regulatory pathways involved in cellular metabolic activity. Tirzepatide research studies typically evaluate how simultaneous stimulation of these receptor systems alters signaling proteins, enzyme activity, and gene expression patterns related to metabolic regulation [2][4].
Additionally, preclinical metabolic models are often used to examine these mechanisms across multiple tissues involved in energy balance, including liver, adipose tissue, and skeletal muscle. These experimental systems allow investigators to observe how incretin-mediated signaling contributes to broader metabolic communication between organs.
Glucose Signaling and Endocrine Pathways
Another important area of tirzepatide research involves endocrine signaling pathways associated with glucose-responsive hormone systems. Incretin hormones normally act as signaling molecules that coordinate nutrient sensing with endocrine responses in pancreatic and gastrointestinal tissues.
In laboratory settings, researchers frequently study tirzepatide in isolated pancreatic islets and endocrine cell models to evaluate signaling responses within insulin-producing beta cells and related endocrine populations. These studies typically measure intracellular second messenger signaling, kinase activation pathways, and transcriptional responses associated with glucose-responsive endocrine signaling networks [5].
Because tirzepatide activates both incretin receptors, it provides a useful experimental model for examining how dual receptor stimulation influences endocrine communication between metabolic tissues.
Lipid Metabolism and Adipose Tissue Biology
Tirzepatide has also been used in experimental research examining lipid metabolism and adipose tissue signaling. Adipocytes respond to hormonal signals that regulate lipid storage, fatty acid mobilization, and metabolic communication with other tissues.
In laboratory studies, researchers often use adipocyte cell cultures and animal models to analyze how incretin receptor activation influences molecular pathways involved in lipid metabolism. These experiments evaluate markers such as enzyme activity involved in fatty acid processing and transcriptional regulation of metabolic genes associated with lipid handling [6].
Dual activation of GIP and GLP-1 receptor systems provides a useful framework for studying how incretin signaling interacts with adipose tissue biology and broader metabolic regulatory pathways.
Liver and Cellular Energy Regulation
The liver plays a central role in coordinating metabolic responses to hormonal and nutrient-derived signals. Research involving tirzepatide frequently investigates how incretin receptor signaling interacts with hepatic metabolic pathways.
In experimental liver models, researchers evaluate molecular markers related to glucose metabolism, lipid processing, and cellular energy regulation. These studies often involve analysis of gene expression patterns, metabolic enzyme activity, and signaling pathways that coordinate metabolic responses within hepatic cells.
By studying tirzepatide in hepatic cell systems and animal models, researchers can explore how incretin signaling contributes to the regulation of metabolic pathways across multiple organ systems [7].
Integrated Metabolic Signaling Across Tissues
Because tirzepatide activates two incretin receptor systems simultaneously, it is frequently used to study coordinated signaling across multiple metabolic tissues. In preclinical models, researchers analyze how dual incretin receptor activation influences molecular signaling networks in tissues such as the pancreas, liver, adipose tissue, and skeletal muscle.
These studies typically examine signaling proteins, transcriptional responses, and metabolic biomarkers associated with cellular energy regulation. By comparing responses across different tissues, investigators can better understand how incretin hormone signaling integrates metabolic communication between organs within complex physiological systems.
This type of integrative research approach helps clarify how hormone signaling pathways coordinate metabolic regulation in controlled laboratory environments.
How Tirzepatide Works (Mechanism of Action)
Being a synthetic peptide, tirzepatide functions as a dual agonist of the glucose-dependent insulinotropic polypeptide (GIP) receptor and the glucagon-like peptide-1 (GLP-1) receptor. These receptors belong to the class B family of G protein–coupled receptors involved in metabolic signaling and nutrient-responsive hormone regulation.
In experimental systems, tirzepatide activates both receptor pathways, enabling investigation of coordinated incretin signaling and downstream molecular processes that regulate cellular metabolism, energy balance, and hormone secretion. Because it interacts with two related receptor systems simultaneously, tirzepatide provides a useful model for studying multi-receptor modulation in biochemical assays and preclinical metabolic research models.
Target Engagement
Tirzepatide binds to and activates both the GIP receptor and the GLP-1 receptor, two structurally related receptors that mediate incretin hormone signaling [1]. The peptide sequence of tirzepatide is primarily derived from native GIP, with specific amino acid substitutions that modify receptor affinity and stability in experimental systems.
These structural modifications allow tirzepatide to engage the GIP receptor with high potency while also maintaining agonist activity at the GLP-1 receptor. Upon binding, the peptide induces conformational changes in the receptor that activate intracellular G protein signaling pathways typical of class B G protein–coupled receptors.
Downstream Signaling Pathways
Activation of incretin receptors by tirzepatide initiates intracellular signaling cascades commonly associated with G protein–coupled receptor activation. Binding of the peptide to GIP or GLP-1 receptors stimulates the Gs protein signaling pathway, leading to activation of adenylate cyclase and increased intracellular cyclic AMP levels [3].
Elevated cyclic AMP can subsequently activate protein kinase A and related signaling molecules, which regulate phosphorylation events and transcriptional responses within responsive cells. These signaling pathways are frequently investigated in receptor pharmacology studies to examine how incretin receptor activation influences cellular metabolic signaling networks.
Cellular Effects in Experimental Models
In laboratory studies using cell culture systems and animal models, tirzepatide has been shown to influence multiple biochemical markers associated with incretin receptor signaling. Experimental investigations have reported modulation of intracellular cyclic AMP levels, alterations in gene expression related to metabolic signaling pathways, and changes in cellular metabolic activity in responsive tissues.
Because tirzepatide activates both GIP and GLP-1 receptor systems, it provides a useful research tool for examining receptor cross-talk, signaling bias, and coordinated regulation of metabolic pathways in controlled experimental environments.
Tirzepatide Comparison: Related Research Compounds
Tirzepatide belongs to a growing class of incretin-based compounds that selectively engage metabolic signaling pathways with increasing receptor complexity. The following comparison positions tirzepatide alongside two related research molecules to show how graduated receptor engagement shapes outcomes across pharmacology studies, biochemical assays, and preclinical metabolic models.
| Property | Tirzepatide | Semaglutide | Retatrutide |
| Type | Synthetic peptide analog | Synthetic GLP-1 peptide analog | Synthetic triple-agonist peptide analog |
| Primary Target | GIP receptor and GLP-1 receptor | GLP-1 receptor | GLP-1 receptor, GIP receptor, and glucagon receptor |
| Mechanism Summary | Dual incretin receptor agonist designed to activate both GIPR and GLP-1R signaling pathways in experimental systems | Selective GLP-1 receptor agonist used to study incretin signaling through a single receptor pathway | Triple receptor agonist used to investigate coordinated signaling across GLP-1, GIP, and glucagon receptor systems |
| Mechanistic Focus | Dual incretin signaling, receptor cross-talk, cAMP pathway activation, coordinated endocrine signaling | GLP-1 receptor pharmacology, glucose-responsive signaling, receptor-specific incretin pathway modulation | Multi-pathway metabolic signaling, receptor network integration, glucagon pathway contribution, energy regulation studies |
| Regulatory Category | Research-use compound supplied for laboratory investigation | Research-use compound supplied for laboratory investigation | Research-use compound supplied for laboratory investigation |
| Research Stage | Widely studied in receptor pharmacology and preclinical metabolic research | Widely studied in incretin biology, receptor signaling, and metabolic pathway research | Emerging but increasingly studied in advanced preclinical metabolic and receptor signaling research |
Tirzepatide is structurally derived primarily from the native GIP peptide sequence and modified to activate both GIP and GLP-1 receptors. This dual agonist profile allows researchers to examine how simultaneous incretin receptor activation influences intracellular signaling pathways such as cyclic AMP signaling and metabolic gene regulation.
Semaglutide represents a more selective experimental model because it primarily targets the GLP-1 receptor without concurrent GIP receptor activity. For laboratory studies investigating receptor-specific incretin signaling, semaglutide provides a useful comparison point for evaluating the differences between single-receptor and dual-receptor agonist activity.
Retatrutide extends this research framework further by incorporating glucagon receptor activation in addition to GLP-1 and GIP receptor signaling. This broader receptor engagement allows researchers to investigate how multi-receptor hormone signaling networks interact across metabolic tissues, making it useful for studying complex metabolic signaling pathways and receptor network interactions in experimental models.
Together, these compounds form a useful group of molecular tools for investigating incretin biology, receptor pharmacology, and metabolic signaling mechanisms in controlled laboratory environments.
Tirzepatide Lab Safety & Handling Guidelines
Tirzepatide supplied by New England Biologics should be handled only by trained laboratory personnel using appropriate chemical safety procedures. The compound is typically supplied as a lyophilized peptide to support stability during storage and transport. For long-term storage, lyophilized tirzepatide is commonly maintained at approximately −4 °F (−20 °C) in a sealed container protected from moisture, heat, and direct light. Maintaining stable storage conditions helps preserve peptide structure and analytical purity over extended research use.
Following reconstitution, peptide solutions are generally stored under refrigerated conditions between 36–46 °F (2–8 °C). Researchers should follow established peptide handling practices to preserve molecular integrity and minimize degradation during experimental workflows.
Handling Guidelines
Laboratory personnel should follow standard peptide handling procedures when working with tirzepatide:
- Store lyophilized vials at −4 °F (−20 °C) or below in a dry, light-protected environment
- Allow the vial to reach room temperature before opening to prevent condensation
- Avoid prolonged exposure to heat, humidity, or direct light during handling
- Use sterile laboratory equipment when preparing experimental solutions
- Minimize repeated freeze–thaw cycles to maintain peptide stability
- Clearly label reconstituted samples with preparation date and concentration
Reconstitution Guidelines
Reconstitution methods may vary depending on experimental requirements, but typical laboratory practices include:
- Reconstitute the lyophilized peptide with sterile bacteriostatic water or an appropriate laboratory buffer
- Introduce the solvent slowly along the inside wall of the vial to reduce foaming
- Allow the solution to dissolve gently without vigorous agitation or vortexing
- Swirl the vial gently until the peptide is fully dissolved
- Store reconstituted solutions at 36–46 °F (2–8 °C) for short-term experimental use
- Prepare aliquots where appropriate to reduce repeated freeze–thaw exposure
Laboratory Safety Protocols
When working with tirzepatide or similar research peptides, standard laboratory safety practices should be followed:
- Wear appropriate personal protective equipment including gloves, laboratory coat, and protective eyewear
- Handle all compounds within approved laboratory workspaces following institutional safety policies
- Avoid inhalation, ingestion, or direct contact with research materials
- Dispose of unused materials and laboratory waste according to institutional chemical disposal guidelines
- Maintain documentation and labeling for all stored research compounds
All products supplied by New England Biologics are intended strictly for laboratory research and development use only. Tirzepatide is not approved for human or veterinary use and must not be introduced into humans or animals under any circumstances.
Frequently Asked Questions
Why do researchers study tirzepatide?
Researchers study tirzepatide because it activates two incretin hormone receptors at the same time: the glucose-dependent insulinotropic polypeptide (GIP) receptor and the glucagon-like peptide-1 (GLP-1) receptor. In laboratory settings, this makes the peptide useful for exploring how incretin hormones influence metabolic signaling, hormone communication between tissues, and intracellular signaling pathways involved in energy regulation.
How should tirzepatide be stored in laboratory settings?
Lyophilized peptide preparations are typically stored at low temperatures in sealed containers to maintain stability and prevent degradation. Many laboratories store research peptides at approximately −4 °F (−20 °C) under dry conditions and protected from light. After reconstitution, solutions are commonly refrigerated and used within established laboratory stability windows. Researchers should follow standard peptide handling practices to maintain sample integrity.
What purity standards does New England Biologics use for research peptides?
Peptides supplied by New England Biologics are produced using controlled synthesis and purification processes designed to achieve high chemical purity suitable for laboratory research. Analytical methods such as chromatography and molecular verification are used to confirm peptide identity and purity, and each batch undergoes quality control testing to support consistency across research applications.
Does New England Biologics provide COA documentation or quality verification?
Yes, all research materials from New England Biologics are typically accompanied by analytical documentation by third-party labs describing purity testing and compound identification. These records help laboratories verify compound identity and maintain reproducibility in experimental workflows.
How quickly does New England Biologics ship research compounds?
Shipping timelines depend on inventory status and destination, but most research orders are processed promptly once confirmed (usually 2-3 days within the US with priority shipping). New England Biologics prioritizes careful packaging and handling to ensure that temperature-sensitive materials arrive in suitable condition for laboratory use.
How is pricing determined for research compounds?
Pricing for research compounds generally reflects synthesis complexity, peptide length or molecular structure, purification requirements, and batch production considerations. Compounds such as tirzepatide require specialized peptide synthesis and purification processes, which influence manufacturing cost and final pricing for laboratory supply. New England Biologics delivers research compounds with high purity and dependable quality, helping laboratories obtain reliable materials while maintaining cost efficiency.
What makes tirzepatide useful for studying incretin hormones?
Tirzepatide activates two related incretin hormone receptors instead of just one. Because of this, researchers can use it to explore how these hormone systems work together in metabolic signaling. In experimental models, this helps scientists study how incretin pathways interact and how hormone signals are coordinated between different tissues.
Regulatory & Legal (U.S.)
All products supplied by New England Biologics are intended strictly for research and development use only. These materials are provided for laboratory investigation and scientific experimentation and are not supplied for use in humans or animals.
This product is not a drug, food, dietary supplement, medical device, or cosmetic. It is not approved by the U.S. Food and Drug Administration (FDA) for medical, diagnostic, or therapeutic use. Any statements regarding the compound are based on published scientific research and have not been evaluated by the FDA. These materials are not intended to diagnose, treat, cure, or prevent any disease.
Materials supplied by New England Biologics must be handled only by qualified professionals trained in laboratory research procedures. The introduction of this product into humans or animals is strictly prohibited and may violate applicable laws and regulations.
Researchers and institutions are responsible for ensuring that all use, storage, handling, and disposal of research materials complies with applicable federal, state, and local regulations, as well as institutional policies governing laboratory research.
Sources & References
- Min T, Bain SC. The Role of Tirzepatide, Dual GIP and GLP-1 Receptor Agonist, in the Management of Type 2 Diabetes: The SURPASS Clinical Trials. Diabetes Therapy. 2021 Jan;12(1):143–157. https://doi.org/10.1007/s13300-020-00981-0
- Qiao Y, Zhou F, Mao T, Gao L. Revitalizing GIP: Therapeutic Potential in Metabolic and Neurodegenerative Disorders. Diabetes, Metabolic Syndrome and Obesity: Targets and Therapy. 2026 Feb 10;19:559–587. https://doi.org/10.2147/DMSO.S559587
- Rees TA, Buttle BJ, Tasma Z, Yang SH, Harris PWR, Walker CS. Tirzepatide, GIP(1-42) and GIP(1-30) Display Unique Signaling Profiles at Two Common GIP Receptor Variants, E354 and Q354. Frontiers in Pharmacology. 2024 Oct 11;15:1463313. https://doi.org/10.3389/fphar.2024.1463313
- Nauck MA, D'Alessio DA. Tirzepatide, a Dual GIP/GLP-1 Receptor Co-agonist for the Treatment of Type 2 Diabetes with Unmatched Effectiveness Regarding Glycaemic Control and Body Weight Reduction. Cardiovascular Diabetology. 2022 Sep 1;21(1):169. https://doi.org/10.1186/s12933-022-01604-7
- Hamamoto Y, Oura T, Hirase T. Insulin Sensitivity and Beta-Cell Function Following Tirzepatide in Japanese Patients with Type 2 Diabetes: A SURPASS J-mono Analysis. Diabetes Therapy. 2025 Apr;16(4):717–729. https://doi.org/10.1007/s13300-025-01704-z
- Lv X, Wang H, Chen C, Zhao Y, Li K, Wang Y, Wang L, Fu S, Liu J. The Effect of Tirzepatide on Weight, Lipid Metabolism and Blood Pressure in Overweight/Obese Patients with Type 2 Diabetes Mellitus: A Systematic Review and Meta-Analysis. Diabetes, Metabolic Syndrome and Obesity: Targets and Therapy. 2024 Feb 12;17:701–714. https://doi.org/10.2147/DMSO.S443396
- Liang J, Liu H, Lv G, Chen X, Yang Z, Hu K, Sun H. Exploring the Molecular Mechanisms of Tirzepatide in Alleviating Metabolic Dysfunction-Associated Fatty Liver in Mice Through Integration of Metabolomics, Lipidomics, and Proteomics. Lipids in Health and Disease. 2025 Jan 10;24(1):8. https://doi.org/10.1186/s12944-024-02416-2
Tirzepatide Structure
Chemical Structure
2D Structure

3D Structure

Chemical Properties
| Amino Acid Sequence | YE-Aib-GTFTSDYSI-Aib-LDKIAQ (C20 fatty acid) AFVQWLIAGGPSSGAPPPS Note: Aib is a non-coded (non-proteinogenic) amino acid – H,H-C(CH₃)₂-COOH |
|---|---|
| Molecular Formula | C225H348N56O68 |
| Molecular Weight | 4813.527 g/mol |
| CAS Number | 2023788-19-2 |
| PubChem CID | 156588324 |
| Synonyms | Tirzepatide, P1206, LY3298176, Dual GIP and GLP-1 receptor agonist (GIP/GLP-1RA) |
| IUPAC Name |
(2S)-2-[[20-[[(5S)-6-[[(2S,3S)-1-[[(2S)-1-[[(2S)-5-amino-1-[[(2S)-6-amino-1-[[(2S)-1-[[(2S)-1-[[(2S)-1-[[(2S)-5-amino-1-[[(2S)-1-[[(2S)-1-[[(2S,3S)-1-[[(2S)-1-[[2-[[2-[(2S)-2-[[(2S)-1-[[(2S)-1-[[2-[[(2S)-1-[(2S)-2-[(2S)-2-[(2S)-2-[[(2S)-1-amino-3-hydroxy-1-oxopropan-2-yl]carbamoyl]pyrrolidine-1-carbonyl]pyrrolidine-1-carbonyl]pyrrolidin-1-yl]-1-oxopropan-2-yl]amino]-2-oxoethyl]amino]-3-hydroxy-1-oxopropan-2-yl]amino]-3-hydroxy-1-oxopropan-2-yl]carbamoyl]pyrrolidin-1-yl]-2-oxoethyl]amino]-2-oxoethyl]amino]-1-oxopropan-2-yl]amino]-3-methyl-1-oxopentan-2-yl]amino]-4-methyl-1-oxopentan-2-yl]amino]-3-(1H-indol-3-yl)-1-oxopropan-2-yl]amino]-1,5-dioxopentan-2-yl]amino]-3-methyl-1-oxobutan-2-yl]amino]-1-oxo-3-phenylpropan-2-yl]amino]-1-oxopropan-2-yl]amino]-1-oxohexan-2-yl]amino]-1,5-dioxopentan-2-yl]amino]-1-oxopropan-2-yl]amino]-3-methyl-1-oxopentan-2-yl]amino]-5-[[(2S)-2-[[(2S)-2-[[2-[[(2S,3S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S,3R)-2-[[(2S)-2-[[(2S,3R)-2-[[2-[[(2S)-2-[[2-[[(2S)-2-amino-3-(4-hydroxyphenyl)propanoyl]amino]-2-methylpropanoyl]amino]-4-carboxybutanoyl]amino]acetyl]amino]-3-hydroxybutanoyl]amino]-3-phenylpropanoyl]amino]-3-hydroxybutanoyl]amino]-3-hydroxypropanoyl]amino]-3-carboxypropanoyl]amino]-3-(4-hydroxyphenyl)propanoyl]amino]-3-hydroxypropanoyl]amino]-3-methylpentanoyl]amino]-2-methylpropanoyl]amino]-4-methylpentanoyl]amino]-3-carboxypropanoyl]amino]-6-oxohexyl]amino]-20-oxoicosanoyl]amino]-5-[2-[2-[2-[2-[2-(carboxymethoxy)ethoxy]ethylamino]-2-oxoethoxy]ethoxy]ethylamino]-5-oxopentanoic acid
|
| InChIKey | AAPYRSPHYSKGIS-MCNPHUAVSA-N |
Sources and References
<h2><b>Sources & References</b></h2>
<ol>
<li>Min T, Bain SC. The Role of Tirzepatide, Dual GIP and GLP-1 Receptor Agonist, in the Management of Type 2 Diabetes: The SURPASS Clinical Trials. Diabetes Therapy. 2021 Jan;12(1):143–157. <a href="https://doi.org/10.1007/s13300-020-00981-0">https://doi.org/10.1007/s13300-020-00981-0</a></li>
<li>Qiao Y, Zhou F, Mao T, Gao L. Revitalizing GIP: Therapeutic Potential in Metabolic and Neurodegenerative Disorders. Diabetes, Metabolic Syndrome and Obesity: Targets and Therapy. 2026 Feb 10;19:559–587. <a href="https://doi.org/10.2147/DMSO.S559587">https://doi.org/10.2147/DMSO.S559587</a></li>
<li>Rees TA, Buttle BJ, Tasma Z, Yang SH, Harris PWR, Walker CS. Tirzepatide, GIP(1-42) and GIP(1-30) Display Unique Signaling Profiles at Two Common GIP Receptor Variants, E354 and Q354. Frontiers in Pharmacology. 2024 Oct 11;15:1463313. <a href="https://doi.org/10.3389/fphar.2024.1463313">https://doi.org/10.3389/fphar.2024.1463313</a></li>
<li>Nauck MA, D'Alessio DA. Tirzepatide, a Dual GIP/GLP-1 Receptor Co-agonist for the Treatment of Type 2 Diabetes with Unmatched Effectiveness Regarding Glycaemic Control and Body Weight Reduction. Cardiovascular Diabetology. 2022 Sep 1;21(1):169. <a href="https://doi.org/10.1186/s12933-022-01604-7">https://doi.org/10.1186/s12933-022-01604-7</a></li>
<li>Hamamoto Y, Oura T, Hirase T. Insulin Sensitivity and Beta-Cell Function Following Tirzepatide in Japanese Patients with Type 2 Diabetes: A SURPASS J-mono Analysis. Diabetes Therapy. 2025 Apr;16(4):717–729. <a href="https://doi.org/10.1007/s13300-025-01704-z">https://doi.org/10.1007/s13300-025-01704-z</a></li>
<li>Lv X, Wang H, Chen C, Zhao Y, Li K, Wang Y, Wang L, Fu S, Liu J. The Effect of Tirzepatide on Weight, Lipid Metabolism and Blood Pressure in Overweight/Obese Patients with Type 2 Diabetes Mellitus: A Systematic Review and Meta-Analysis. Diabetes, Metabolic Syndrome and Obesity: Targets and Therapy. 2024 Feb 12;17:701–714. <a href="https://doi.org/10.2147/DMSO.S443396">https://doi.org/10.2147/DMSO.S443396</a></li>
<li>Liang J, Liu H, Lv G, Chen X, Yang Z, Hu K, Sun H. Exploring the Molecular Mechanisms of Tirzepatide in Alleviating Metabolic Dysfunction-Associated Fatty Liver in Mice Through Integration of Metabolomics, Lipidomics, and Proteomics. Lipids in Health and Disease. 2025 Jan 10;24(1):8. <a href="https://doi.org/10.1186/s12944-024-02416-2">https://doi.org/10.1186/s12944-024-02416-2</a></li>
</ol>